Unit 3: The atmosphere and hydrosphere: learn about composition, functions of the atmosphere, the water cycle, the water molecule (solid, liquid or vapor), weather and climate.
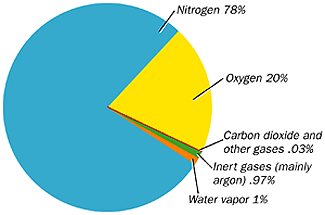
Composition: 78% Nitrogen, 21% Oxygen, and the rest is composed of water vapour, CO2, CH4, dust particles, pollen, etc.
90% of the atmosphere's mass is located closer to the surface and the rest is spread in a wide space (500 km). The first layer of the atmosphere, the Troposphere contains 90% of the atmosphere's mass.
Find here the atmosphere's structure and the molecule of water.
The atmosphere's functions:
1.- To regulate temperature and climate.
2.- To protect the Earth.
3.- To contain vital gases for living organisms.
4.- It's the place for weather phenomena.
The hydrosphere's functions:
1.- To solve substances.
2.- To support life.
3.- To transport substances.
4.- To regulate temperature.

Let me introduce the MIcky mouse molecule: H2O !!!
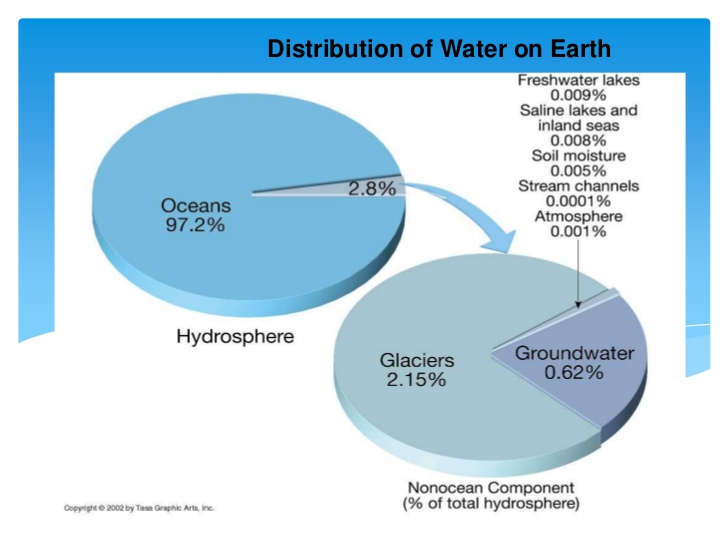
Salt water: more than 35 grams of salts per liter of water.
Fresh water: less than 35 grams of salts for every liter of water.
Now you know the water molecule and its Micky mouse structure. Let's study its properties.
Properties of water:
1.- Water is a good solver (it dissolves mostly of the substances, but fats)
2.- Water has a high specific heat (that means that it needs a big amount of energy to rise its temperature one degree). Water regulates temperature
3.- Water transports substances.
4.- Ice is less dense than liquid water.
5.- It participates in chemical reactions.
6.- Water molecules display high adhesive capacity between them and other substances.
What if water didn't have its properties? Guess what would happen.....
Salt water: more than 35 grams of salts per liter of water.
Fresh water: less than 35 grams of salts for every liter of water.
Now you know the water molecule and its Micky mouse structure. Let's study its properties.
Properties of water:
1.- Water is a good solver (it dissolves mostly of the substances, but fats)
2.- Water has a high specific heat (that means that it needs a big amount of energy to rise its temperature one degree). Water regulates temperature
3.- Water transports substances.
4.- Ice is less dense than liquid water.
5.- It participates in chemical reactions.
6.- Water molecules display high adhesive capacity between them and other substances.
What if water didn't have its properties? Guess what would happen.....
| laboratory_boiling_water.docx | |
| File Size: | 12 kb |
| File Type: | docx |
| wondering-about-water.pdf | |
| File Size: | 38 kb |
| File Type: | |
Here you can find a presentation on the atmosphere and hydrosphere.
| atmospherehydrosphere_1.pptx | |
| File Size: | 1710 kb |
| File Type: | pptx |
Read the following files ot learn about matter, states and pressure.
| states_of_matter_grid.docx | |
| File Size: | 11 kb |
| File Type: | docx |
| difference_between_evaporation_and_boiling.docx | |
| File Size: | 17 kb |
| File Type: | docx |
GAS APPLET LABORATORY
The following game is a model that explains the behavour of gas molecules under certain conditions of pressure, temperature and volume. We will play with it to explain the reasons for evaporation and condensation.
About clouds:
| whats-in-a-cloud.pdf | |
| File Size: | 198 kb |
| File Type: | |
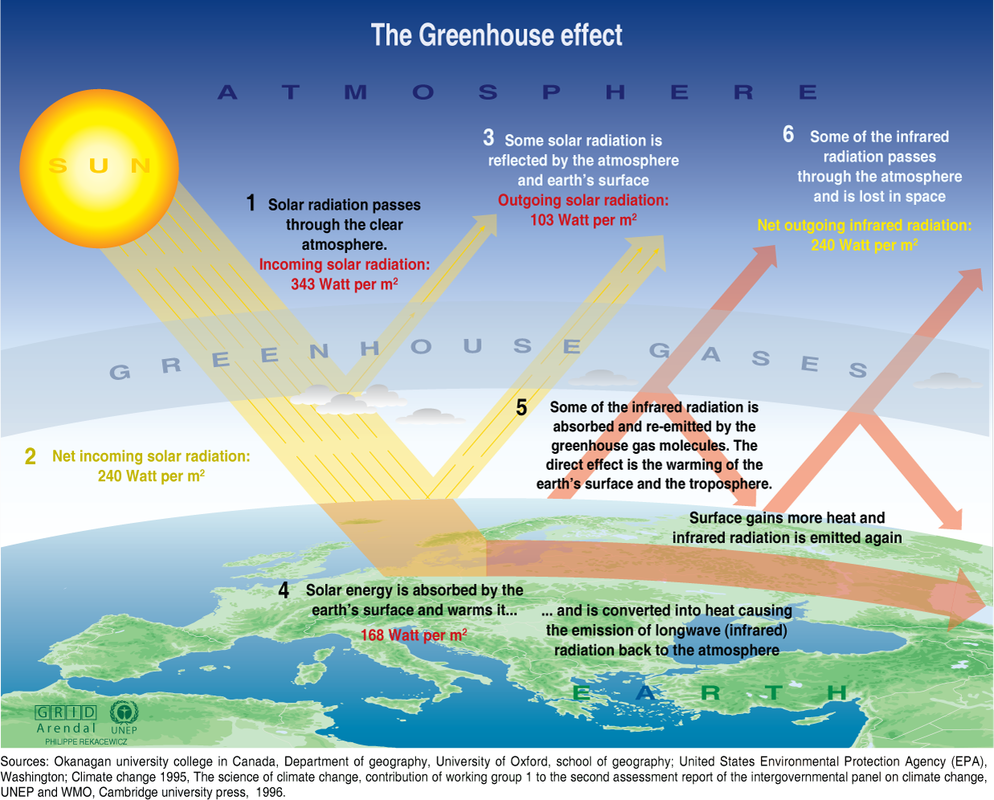
THE GREENHOUSE LAYER AND THE OZONE LAYER
The greenhouse effect is both good and bad for the ecosystem. The greenhouse effect layer keeps the heat under the atmosphere heating it and allowing the average of 15 ºC of temperature. But when the layer is too thick (it contains many gases) it keeps too much heat and the temperature rises producing the global warming.
Ozone, a friend or a foe?
| ozone-friend-and-foe.pdf | |
| File Size: | 994 kb |
| File Type: | |
The Federal USA Goverment has created this page to learn about climate change and the ways you have to reduce it.
Water pollution and water cleaning:
Things you can do to conserve water:
| water-conservation.pdf | |
| File Size: | 547 kb |
| File Type: | |